DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
MannKind Corporation (Nasdaq: $MNKD): Ralinepag DPI, Tyvaso/Tresmi, Afrezza and the Next Catalyst-Rich Chapter Updated

MannKind is not a clean story. That is precisely why it deserves a deep dive. This is a company with one of the most tortured histories in small-cap biotech: years of hope around Afrezza, a painful Sanofi breakup, repeated market skepticism, debt and dilution concerns, and a long struggle to prove that inhaled therapeutics could become more than a scientific idea. Yet MannKind is still here, and the current version of the company is meaningfully different from the old single-product Afrezza story.
Recursion Pharmaceuticals (Nasdaq: $RXRX): Q1 2026 turns the AI-biotech story into an execution test

Recursion Pharmaceuticals did not report a “commercial” quarter in the traditional biotech sense. There is still no approved product, no drug-sales revenue and no recurring product line that can be valued like a de-risked commercial-stage company. But Q1 2026 still matters because it updates the three pillars of the RXRX story: the AI-powered platform is generating real clinical programs, the company is reducing burn versus 2025, and lead asset REC-4881 in familial adenomatous polyposis is moving into the stage where FDA alignment becomes the key inflection point.
$SUPN, $RLAY and $ADPT: Three Q1 2026 Earnings Reports, Three Different Biotech Risk Profiles
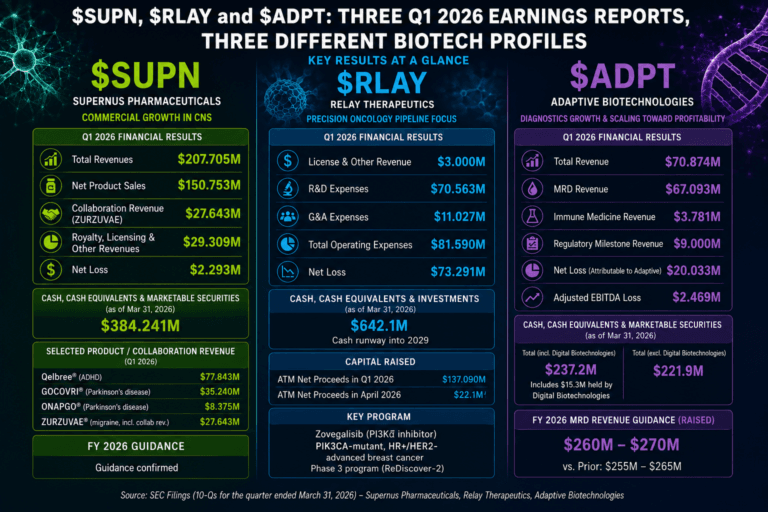
This report looks at three Q1 2026 biotech earnings releases that are worth grouping together because they show three very different types of healthcare equity risk. Supernus Pharmaceuticals is a commercial CNS company trying to scale growth products while absorbing spending and legacy erosion. Relay Therapeutics is a clinical-stage precision oncology company with a long cash runway, but still heavy operating losses. Adaptive Biotechnologies is a commercial diagnostics company where MRD growth and clonoSEQ volume are the central story.
Rigel Pharmaceuticals (Nasdaq: $RIGL): Q1 2026 Profitability, the Lilly Reset and the Harder Test for a Commercial Biotech Turnaround

Why This Deep Dive Matters Now
The timing is what makes Rigel worth a fresh, event-driven deep dive. The company entered 2026 after a much stronger 2025, with record net product sales, full-year profitability and a larger cash position. That already made the stock more relevant for small-cap biotech investors searching for companies with real revenue rather than purely clinical speculation.
Then came the Eli Lilly termination. That changed the framing immediately. Before the termination, Rigel could be described as a commercial-stage biotech with three marketed products and a potentially meaningful partnered RIPK1 program backed by one of the most important pharmaceutical companies in the world. After the termination, investors have to remove part of that long-term optionality from the story.
Verrica Pharmaceuticals: VP-315 Phase 2 Data Put a Fresh Spotlight on Basal Cell Carcinoma

Verrica Pharmaceuticals returned to the biotech watchlist on May 5 with a clinically interesting update on VP-315 / ruxotemitide, its investigational oncolytic peptide program in basal cell carcinoma. The headline is not merely that the company will present at the 2026 Society for Investigative Dermatology Annual Meeting in Chicago. The important part is that Verrica is highlighting Phase 2 data showing tumor reductions in lesions that were not directly injected with VP-315.
Ocugen (Nasdaq: $OCGN): Q1 2026, Convertible Financing, Pipeline Catalysts and the New Risk/Reward Map

Ocugen has just reset the financial framework around its equity story. The company entered 2026 as a high-beta, pre-commercial biotech with a pipeline that had visible clinical and regulatory milestones but a balance sheet that still looked too narrow for the size of the opportunity it was trying to pursue. The May 2026 financing changes that discussion. Ocugen priced $115 million aggregate principal amount of 6.75% convertible senior notes due 2034, with an option for the initial purchaser to acquire up to an additional $15 million. The company expects the transaction to close on May 7, 2026, subject to customary conditions, and expects net proceeds of roughly $99.5 million before the optional additional notes. A portion of the proceeds, approximately $32.7 million, is expected to be used to fully repay the Avenue Capital debt facility, including principal, accrued and unpaid interest, a prepayment fee, and related expenses.
Oculis Holding AG (Nasdaq: $OCS): the June 2026 DIAMOND readout is the real test
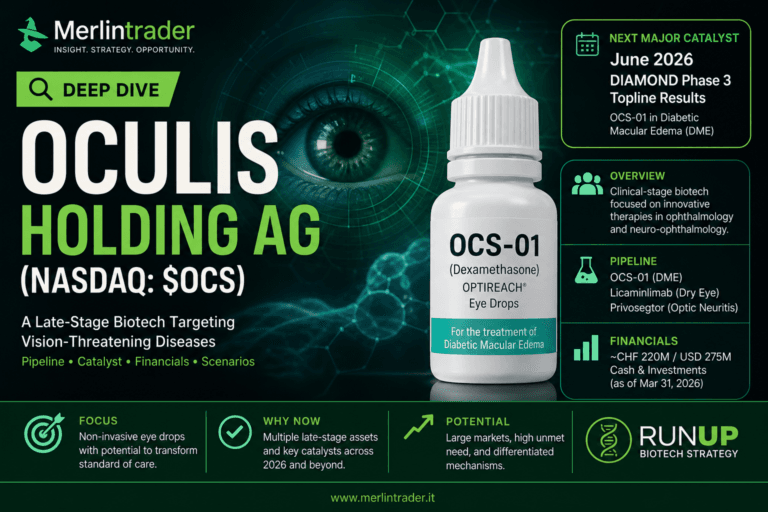
Oculis Holding AG is not a generic small biotech waiting for a vague future update. It is a late-stage ophthalmology and neuro-ophthalmology company approaching a defined Phase 3 readout in diabetic macular edema, one of the most commercially relevant retina indications in eye care.
The core of the story is OCS-01, a high-concentration dexamethasone eye drop built on the company’s OPTIREACH® formulation technology. The thesis is easy to understand but difficult to prove: if a topical eye drop can deliver clinically meaningful benefit in a posterior-segment disease such as diabetic macular edema, Oculis could open a new therapeutic category in a market still dominated by intravitreal injections and invasive treatment pathways.
Ocular Therapeutix (Nasdaq: $OCUL): Sanofi Rumor, AXPAXLI and the Real Takeover Logic Behind the Wet AMD Story

Ocular Therapeutix is back in focus after media reports linked Sanofi to a possible takeover approach. According to reports citing French publication La Lettre, Sanofi previously approached Ocular with an offer around $16 per share, Ocular’s board reportedly rejected that initial proposal, and Sanofi may be considering or preparing a higher bid. Neither Ocular Therapeutix nor Sanofi has confirmed a transaction, and that distinction matters. The confirmed foundation of the story is not the takeover rumor; it is AXPAXLI, Ocular’s investigational axitinib intravitreal hydrogel implant, which recently produced positive Phase 3 SOL-1 data in wet age-related macular degeneration.
Rezolute, Inc. (Nasdaq: $RZLT): sunRIZE is not rescued yet — but the PES data make the FDA-package story harder to ignore

Rezolute’s latest update is important because it gives the congenital hyperinsulinism story a second act. It does not erase the first act. The first act remains the December 2025 disclosure that the Phase 3 sunRIZE trial of ersodetug missed its primary endpoint and its key secondary endpoint. That failure damaged the clean registration narrative and forced the company into a more complex argument: the drug may have shown meaningful biological and clinical activity, but the trial’s primary endpoint may have been confounded by behavioral and measurement issues tied to self-monitored blood glucose.
Fate Therapeutics (Nasdaq: $FATE): iPSC Cell Therapy, FT819 and the High-Stakes Autoimmune CAR-T Reset Updated 04 may 2026

Fate Therapeutics is one of the more interesting “fallen platform” stories in biotech. The company is not a single-asset microcap built around one binary Phase 2 readout; it is a long-running cell therapy platform company trying to prove that induced pluripotent stem cells, or iPSCs, can support standardized, off-the-shelf cellular medicines across autoimmune disease and oncology. The market, however, is no longer paying a premium for platform ambition alone. After several difficult years for allogeneic and off-the-shelf cell therapy names, investors want evidence: durable clinical activity, manageable safety, realistic manufacturing economics, regulatory clarity and a capital structure that does not punish existing shareholders before the platform has time to mature.
Summit Therapeutics ( $SMMT ): the HARMONi-3 selloff does not kill the story, but it changes the burden of proof

Summit Therapeutics is not a normal quarterly earnings story. The company has no commercial revenue profile to dissect in the usual way, no product-sales margin trend to model, and no near-term EPS narrative that matters more than its clinical and regulatory path. $SMMT is a concentrated bet on ivonescimab, also known as SMT112 in Summit’s licensed territories, a PD-1/VEGF bispecific antibody that has become one of the most closely watched potential challengers to the PD-1 oncology establishment.
Ardelyx Inc. (Nasdaq: $ARDX): The Growth Story Is No Longer Theoretical

Ardelyx is now a commercial-stage biotech where the core debate has moved from “can the product sell?” to “can the company turn rapid product growth into durable operating leverage?” That shift matters. It means $ARDX is no longer trading purely as a binary FDA story or early-launch hope. It is trading as a real revenue company with two approved U.S. products, a differentiated NHE3 biology platform, a growth engine in IBSRELA, a more complicated but still relevant kidney franchise in XPHOZAH, and a pipeline extension strategy that could make today’s commercial infrastructure more valuable over time.
Contineum Therapeutics (Nasdaq: $CTNM): PIPE-791 Puts Chronic Pain Back on the Map

Contineum Therapeutics is a clinical-stage biotechnology company focused on oral small molecules for neuroscience, inflammation and immunology, a space the company abbreviates as NI&I. The stock is back in focus because Contineum reported positive topline data from an exploratory Phase 1b trial of PIPE-791 in chronic pain on April 30, 2026. The market setup is simple: a small biotech with a relatively concentrated pipeline just produced fresh human data in a high-interest indication, while already carrying a separate Phase 2 IPF program and a partnered Johnson & Johnson asset in depression.
Axsome Therapeutics (Nasdaq: $AXSM): AUVELITY Approval Changes the Alzheimer’s Agitation Story

Axsome is no longer just a high-growth CNS name with a pending binary catalyst. After the FDA approval of AUVELITY for agitation associated with dementia due to Alzheimer’s disease, the debate shifts to launch execution, payer access, physician adoption, label positioning and whether the market has already priced in a large part of the upside.
Spyre Therapeutics 2026 (Nasdaq: $SYRE): When Great Clinical Data Matter More Than Promises

Spyre Therapeutics is now one of the more important clinical-stage IBD stories on the U.S. biotech radar. The company is not commercial, it has no approved product, and it remains a high-risk development-stage name. But after the April 13, 2026 SPY001 Phase 2 SKYLINE-UC Part A induction data, the market has a real clinical datapoint to underwrite rather than a pure slide-deck narrative.
Revelation Biosciences, Inc. (Nasdaq: $REVB): Gemini, AKI, CKD and the High-Risk Micro-Cap Repricing Setup

Revelation Biosciences, Inc. (NASDAQ: REVB) is a clinical-stage biopharmaceutical company based in San Diego, California, focused on developing therapies designed to modulate the innate immune system. Its lead product candidate, Gemini, is a proprietary intravenous formulation of PHAD® (phosphorylated hexaacyl disaccharide), a Toll-like receptor 4 agonist intended to rebalance innate immune activity and reduce organ damage associated with acute and chronic inflammatory stress.
Viking Therapeutics (Nasdaq: $VKTX): the May 2026 Deep Dive

VK2735 has moved from promise to execution: two subcutaneous Phase 3 VANQUISH trials are fully enrolled, maintenance-dose data are expected in Q3 2026, oral Phase 3 development is now guided for Q4 2026, and the Q1 update confirms both the opportunity and the spending intensity of Viking’s obesity strategy.
XTL Biopharmaceuticals Ltd. $XTLB: Psyga Bio Deal Turns a Tiny IP Portfolio Story Into a Psychedelic Biotech Catalyst Trade

XTL Biopharmaceuticals has signed a definitive agreement to acquire Psyga Bio, an Israeli psychedelic and functional mushroom platform with a licensed GMP-ready manufacturing facility, a proprietary mushroom-strain library and seven approved Phase 2a human clinical trials. The opportunity is obvious. So are the risks: shareholder approval, Nasdaq listing status, interested-party governance, liquidity, dilution and the need to convert an exciting headline into real clinical execution.
Teva Pharmaceutical Industries (NYSE: $TEVA): Q1 2026 Turns the Pivot to Growth Into a Harder-to-Ignore Story

Teva’s Q1 2026 report is important because it compresses the entire turnaround debate into one clean question: is this still a heavily indebted generics company trying to defend mature assets, or has it become a hybrid biopharma platform where innovative medicines, biosimilars and late-stage pipeline optionality can finally outweigh the drag from legacy generics?
Aclaris Therapeutics (Nasdaq: $ACRS): ATI-052 Phase 1a Data, ATI-2138 Lichen Planus Pivot and the 2026 Catalyst Reset

Aclaris Therapeutics is back on the biotech catalyst radar because the April 28, 2026 update gives the market two things it was waiting for: more complete first-in-human data for ATI-052 and a clearer development path for ATI-2138. The new story is not a single-asset binary event. It is a pipeline reset around immuno-inflammatory diseases, where Aclaris is trying to build a differentiated position with one long-acting biologic and one oral T-cell pathway inhibitor