DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
Microbot Medical (Nasdaq: $MBOT): First LIBERTY Revenue, Real Doctors and the Small-Cap Robotics Test
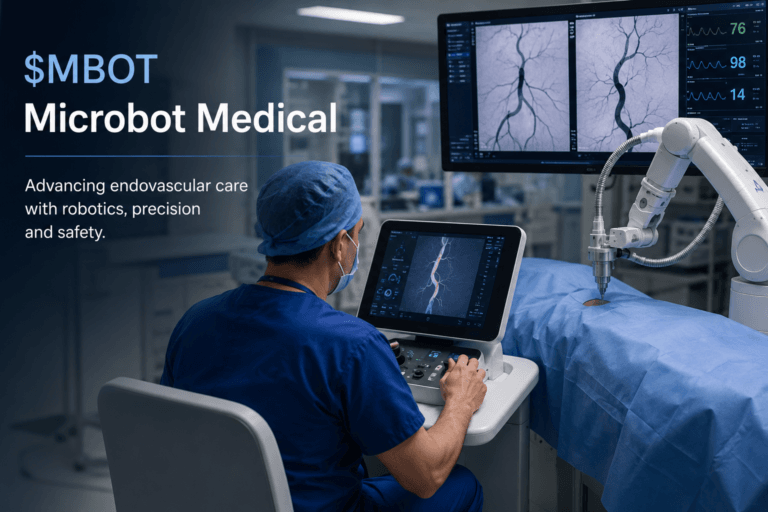
Microbot Medical is not a clean “robotics stock” headline. It is a much more interesting, messier and more human story: a small public medical device company trying to commercialize a remotely operated, single-use robotic system for endovascular procedures, while the market debates whether the first commercial signs are meaningful enough to justify the risk. The company has now reported first-quarter 2026 revenue tied to the limited market release of LIBERTY, says Q2 revenue had already exceeded Q1 revenue by mid-quarter, and has scheduled a May 18 investor call where real physician users of the system are expected to join management.
Viral Threats, Liver Fibrosis and Muscle Regeneration: $TRAW, $GALT and $MSLE Add Another Layer to the May 15 Biotech Tape

Merlintrader Biotech Update Published / Aggiornato: May 15, 2026 $TRAWDaily chart $GALTDaily chart $MSLEDaily chart $TRAW $GALT $MSLE Biotech May 15 Updates Viral Threats, Liver Fibrosis and Muscle Regeneration: $TRAW, $GALT and $MSLE Add Another Layer to the May 15…
Regulatory Paths, PDUFA Risk and Pivotal Oncology: $ATYR, $BTAI and $MBRX After the May 15 Updates

The final May 15 biotech group is not built around early proof-of-concept excitement. It is built around regulatory execution, pivotal-stage risk and the constant question that follows small and mid-sized biotech companies: can the next regulatory or clinical milestone arrive before financing pressure becomes the dominant market narrative? aTyr Pharma, BioXcel Therapeutics and Moleculin Biotech all issued updates on May 15 that give traders and long-form biotech readers a concrete catalyst map, but each company also comes with a very different risk profile.
Alzheimer’s, RAS Oncology and iNKT Cell Therapy: $ANVS, $IMRX and $INKT Enter the May 15 Biotech Watchlist

The May 15 biotech tape delivered another group of earnings and business updates that deserve more than a quick headline scan. Annovis Bio pushed its neurodegenerative disease story forward with a Phase 3 Alzheimer’s update, 85% enrollment in its pivotal Alzheimer’s disease trial, a positive DSMB safety recommendation, a Parkinson’s open-label extension study now 40% enrolled, and an outlined NDA path for buntanetap. Immuneering reported a very different type of update, centered on atebimetinib in pancreatic cancer, an upcoming ASCO oral presentation, a pivotal Phase 3 MAPKeeper 301 trial now recruiting, and a strong cash position of $198.6 million expected to fund operations into 2029. MiNK Therapeutics added a smaller but clinically interesting cell-therapy angle, advancing its off-the-shelf iNKT platform into randomized Phase 2 validation in severe acute lung injury and critical illness, with additional clinical data scheduled for ATS on May 20.
Clinical Signals, Commercial Execution and Regulatory Paths: $TNYA, $SPRY and $BCDA After the May 15 Updates
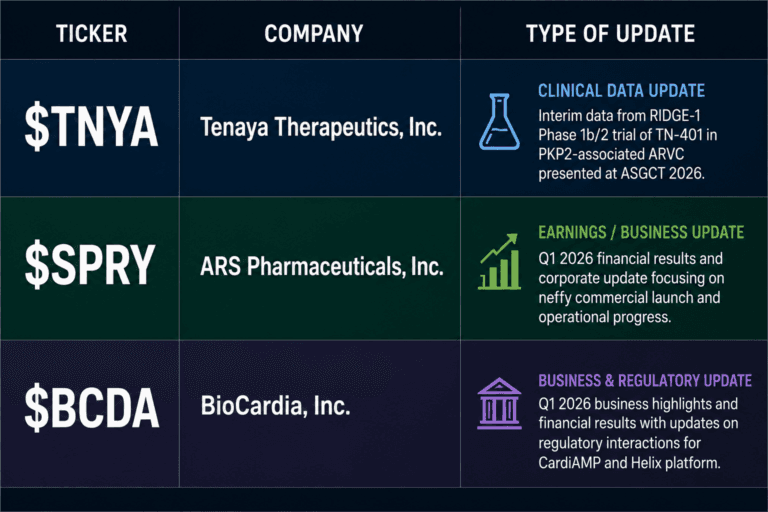
Clinical Signals, Commercial Execution and Regulatory Paths: $TNYA, $SPRY and $BCDA After the May 15 Updates
Innate Pharma (Nasdaq: $IPHA): Cancer Immunotherapy, PACIFIC-9, Lacutamab and the 2026 Catalyst Window

Innate Pharma static chart — IPHA Static Finviz image. Referral applies only if the chart is clicked. The image is loaded without an affiliate parameter. The affiliate referral is attached only to the outbound chart link. Merlintrader Stock Hub ·…
Cingulate Inc. (Nasdaq: $CING): Q1 Cash Build, Commercial Readiness and the Final Stretch Toward the May 31 FDA Catalyst

Static Finviz chart. Affiliate referral is attached only to the outbound chart link, not to the image load. Latest Insight · May 14, 2026 Nasdaq: $CING Q1 2026 · FDA Catalyst Cingulate Inc. (Nasdaq: $CING): Q1 Cash Build, Commercial Readiness…
Three Biotech Earnings Reports, Three Different Catalyst Stories: Gene Therapy, Autoimmune Cell Therapy and Oncology in Focus — $RGNX, $CABA, $KPTI
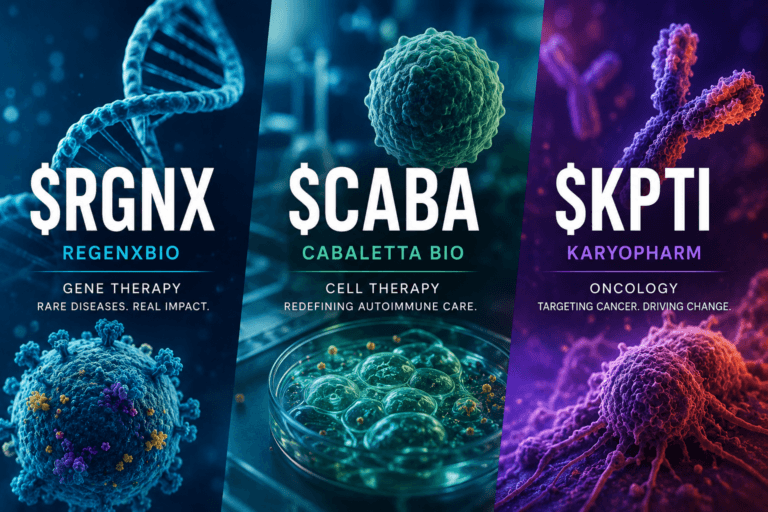
REGENXBIO, Cabaletta Bio and Karyopharm all reported first-quarter 2026 updates on the same day, but the investment debate is not simply about quarterly EPS. The real story is a cluster of catalyst paths: Duchenne gene therapy and AbbVie-partnered retinal disease at REGENXBIO, rese-cel autoimmune cell therapy at Cabaletta, and a selinexor franchise trying to extend beyond multiple myeloma at Karyopharm.
Three Biotech Earnings Stories Moving in Different Directions: $PGEN, $AQST and $ALLO
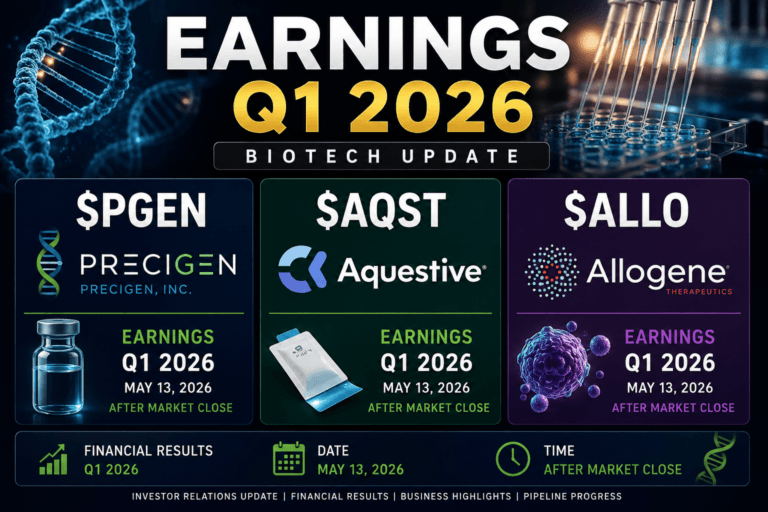
The May 2026 biotech earnings window produced three very different stories in Precigen, Aquestive Therapeutics and Allogene Therapeutics. They belong to the same broad sector, they all sit inside the volatile small-to-mid-cap biotechnology universe, and they all trade with catalyst sensitivity.
Three Biotech Stocks With Fresh May 13 Updates: $TYRA, $SPRB and $PDSB
May 13 delivered another packed biotech earnings and business-update session. For traders, the hard part is not simply finding companies that issued a press release. The hard part is separating routine quarterly updates from updates that actually change the near-term story. The three names in this article — Tyra Biosciences, Spruce Biosciences and PDS Biotech — all fit that second category, but for very different reasons.
Three 2026 FDA Decisions Testing Whether New Drugs Can Beat Imperfect Standards: Unicycive, Protagonist and Savara ($UNCY $PTGX $SVRA)

A PDUFA date is easy to understand. It creates a countdown, gives traders a calendar anchor and makes a biotech story feel tradable. But the FDA date is rarely the whole thesis. In near-approval biotech, the first question is whether the drug can be approved. The second question, often more important for long-term value, is whether the drug can change behavior after approval. That second question is where many apparently clean catalyst stories become complicated.
Viking Therapeutics $VKTX, Structure Therapeutics $GPCR and Altimmune $ALT: The Next Obesity Drug Race

The obesity-drug market has moved from discovery excitement into commercial combat. That is the central change investors have to absorb. In 2023 and 2024, it was still possible to build a strong biotech story around the idea that a company had an obesity asset with meaningful weight-loss data. In May 2026 that is no longer enough. The benchmark is no longer theoretical. Patients can already get injectable semaglutide, injectable tirzepatide, oral semaglutide and oral orforglipron in the U.S. market, while older weight-management drugs still occupy price-sensitive, generic or non-incretin niches. Novo Nordisk's Wegovy pill was approved in December 2025 as an oral GLP-1 option for weight management, and Eli Lilly's Foundayo, orforglipron, was approved in April 2026 as a once-daily oral GLP-1 pill that Lilly says can be taken without food or water restrictions. Those approvals make the next phase more difficult, but also more valuable, because they validate patient demand for convenience and scale. 3 1 2
3 Biotech Stocks Facing Near-Term FDA Catalysts: MannKind ($MNKD), Viridian Therapeutics ($VRDN) and Vera Therapeutics ($VERA)

Biotech does not need to be vague to be speculative. The best watchlist stories usually sit in the middle: visible enough to be researched, near enough to matter, but still uncertain enough to create price discovery. MannKind ($MNKD), Viridian Therapeutics ($VRDN) and Vera Therapeutics ($VERA) fit that profile heading into late May, June and July 2026.
3 Biotech Stocks to Watch: PTC Therapeutics ($PTCT), Enliven Therapeutics ($ELVN) and Edgewise Therapeutics ($EWTX)

Biotech watchlists are most useful when they separate the type of risk behind each ticker. PTC Therapeutics is not the same story as Enliven Therapeutics, and Enliven is not the same story as Edgewise Therapeutics. $PTCT is now a commercial execution and guidance story. $ELVN is a clean oncology catalyst story built around a potential Phase 3 transition in chronic myeloid leukemia. $EWTX is a broader clinical-platform story where investors are watching both cardiac and muscular dystrophy programs for data that could reshape the company’s valuation narrative before the end of 2026.
Cingulate ( $CING ) vs Aytu BioPharma ( $AYTU ): What a Commercial ADHD Peer Can — and Cannot — Tell Us Before CING’s May 31 PDUFA

CING is a pre-commercial FDA catalyst story built around CTx-1301. AYTU is a commercial specialty pharma/CNS story with real ADHD revenue, EXXUA optionality, debt, warrants and execution complexity. The useful comparison is not “which one is better,” but what kind of risk each company represents.
Traws Pharma (Nasdaq: $TRAW): Hantavirus Urgency, Antiviral Pipeline and the High-Risk Reset Story

Traws is trying to reposition itself around clinical-stage antiviral assets for respiratory and outbreak-prone viral threats. The new hantavirus initiative adds a timely public-health narrative, but the investment story still depends on execution around tivoxavir marboxil, ratutrelvir, regulatory risk, financing mechanics and the company’s ability to convert scientific optionality into credible clinical progress.
Atara Biotherapeutics (Nasdaq: $ATRA): FDA Type A Meeting Opens a Narrower Resubmission Path for Tabelecleucel

Atara Biotherapeutics has received the kind of regulatory update that can revive a micro-cap biotech narrative, but only if investors read it with discipline. The May 7, 2026 announcement is not a new approval, not a BLA acceptance, and not a PDUFA date. It is something more technical and, for this specific story, potentially more important: after a Type A meeting, FDA has agreed that a single-arm study using an appropriate historical control, conducted in a pre-specified manner and applicable to the trial population, could serve as an adequate and well-controlled study to support a future marketing application for tabelecleucel, also known as tab-cel.
Humacyte Inc. (Nasdaq: $HUMA): Symvess Is Approved, But the Real Test Is Commercial Execution UPDATED May 13

Humacyte is no longer just a speculative pre-approval biotechnology story. The company has an FDA-approved product, a real commercial launch, hospital ordering activity, international regulatory work, military and trauma relevance, and a near-term Phase 3 dialysis catalyst. That alone makes the story more mature than many small-cap biotech names trading purely on hope.
Ultragenyx Pharmaceutical Inc. (Nasdaq: $RARE): May 2026 Deep Dive

Ultragenyx Pharmaceutical enters May 2026 as a rare-disease platform that has already been through the emotional part of a major reset. The December 2025 failure of setrusumab, also known as UX143, in the Phase 3 ORBIT and COSMIC studies for osteogenesis imperfecta changed the equity story. Before that readout, part of the market still treated UX143 as a potential next commercial pillar. After the readout, that assumption had to be removed or heavily discounted. The drug improved bone mineral density, but it did not deliver statistically significant fracture-rate reductions in the pivotal studies. In rare-disease investing, that distinction matters. Biology can look encouraging; regulators, payers and investors still need clinically persuasive outcomes.
aTyr Pharma Inc. (Nasdaq: $ATYR): The EFZO-FIT Collapse, the Rebuild Trade and the FDA Path That Now Defines the Story

aTyr Pharma is no longer the clean Phase 3 binary story that traders were watching before September 2025. It is now a post-collapse biotech reconstruction trade. The difference matters. Before EFZO-FIT read out, the market could frame ATYR around a simple question: would efzofitimod show a statistically persuasive ability to reduce oral corticosteroid use in pulmonary sarcoidosis? After the readout, the question changed completely. The Phase 3 trial did not meet its primary endpoint. The drug is not de-risked. The company now has to defend the clinical relevance of secondary and supportive signals, persuade regulators that the totality of evidence may still deserve a path forward, protect the balance sheet, and keep investors from reducing the entire story to a failed late-stage asset.