DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category NRXP
NRX Pharmaceuticals Inc
NRx Pharmaceuticals ( $NRXP ) — From FDA Alignment to Presidential Backing
NRx Pharmaceuticals has become a far more layered story than the version many traders were following a few months ago. It is no longer just an abstract ketamine thesis or a speculative real-world-evidence talking point. As of April 20, 2026, the company has three separate but connected tracks worth watching: a near-term generic-drug approval set-up through KETAFREE™, a potentially more valuable but still higher-risk NDA strategy for NRX-100, and an expanding clinic / neurotechnology ecosystem designed to create commercial infrastructure around both drug assets.
NRx Pharmaceuticals ( $NRXP ) After FY2025: A Multi-Asset Story with Regulatory, Commercial, and Strategic Upside
EN IT Merlintrader Deep Dive | NRXP NRx Pharmaceuticals After FY2025: A Multi-Asset Story with Regulatory, Commercial, and Strategic Upside NRx is no longer just a one-line speculative ketamine story. After the March 2026 annual report and earnings call, the…
NRx Pharmaceuticals ( $NRXP ) – FDA Minutes Lock In the NRX-100 NDA Path
NRx Pharmaceuticals finally got the kind of news biotech traders usually wait for: not just another optimistic corporate interpretation, but formal written FDA minutes that document the regulatory path. In today’s release, the company said it received the confirmatory written minutes from its February 17, 2026 in-person Type C meeting with the FDA. According to NRx, those minutes support a path to file an NDA for NRX-100 by June 2026 using existing adequate and well-controlled clinical trial data together with confirmatory Real World Evidence. The company also said the FDA confirmed that no additional clinical trials and no bridging studies are required for the preservative-free formulation
NRX Pharmaceuticals Inc ($NRXP)
NRx Pharmaceuticals is a micro-cap biotech focused on severe psychiatric conditions: suicidal depression, bipolar depression with suicidality, PTSD and related disorders. The core of the story is NRX-100, a preservative-free intravenous ketamine program supported by a large Real-World Evidence dataset, and NRX-101, an oral D-cycloserine + lurasidone combination with Breakthrough designation. Around those assets, the company is building HOPE Therapeutics, a network of interventional psychiatry clinics that should both validate the care model and generate revenue.
NRXP NRX Pharmaceuticals Inc
NRx has licensed a nationwide Real-World Evidence dataset from Osmind covering more than 70,000 patients treated with IV ketamine or intranasal esketamine for major depression with suicidal ideation. The full analysis will be submitted to the FDA in support of an Accelerated Approval application for NRX-100, the preservative-free ketamine IV formulation already under Fast Track Designation for suicidal ideation in depression and bipolar depression.
NRXP and the Phantom PDUFA 2
Over the last 18–24 months, NRx Pharmaceuticals (NRXP) has built a storyline around NRX-100 (preservative-free IV ketamine) that many retail traders translated into a simple, crisp line: “PDUFA by year-end 2025.”
NRXP and the Phantom PDUFA: When ‘By Year-End’ Isn’t a Date
NRX-100 was supposed to be one of those year-end catalysts that keep biotech traders awake at night: fast-track status, fee waivers, a new FDA voucher program, and a decision “by year-end 2025”. Yet, as the calendar runs out, the line between guidance and reality still looks blurry.
NRXP NRX Pharmaceuticals Inc

NRx has announced the conversion of the remaining 5.4M USD owed to Anson Funds into common equity, with no additional warrants or repricing features disclosed. Management states that, following this transaction, the year-end 2025 balance sheet is expected to show no remaining convertible debt and a “debt-free” capital structure.
NRXP

NRx Pharmaceuticals (NRXP) | Investment Research Report ⚠️ IMPORTANT NOTICE / AVVISO IMPORTANTE: This document is a news digest and company profile compiled from publicly available sources (SEC filings, press releases, official company announcements). It is NOT investment advice, NOT…
$NRXP
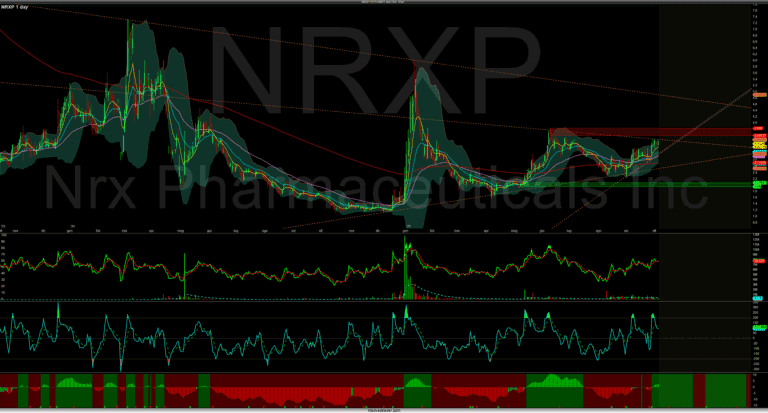
Il grafico è interessante ma non vedo grossi catalyst vicino. Ma mentre si mantiene in trend è buona. Questa è una fase critica per il grafico, vediam se brekka 3,82 intanto. Catalyst attuali / imminenti Non Altri contenuti su NRXP…