DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
TENX Tenax Therapeutics

Tenax Therapeutics is advancing oral levosimendan for pulmonary hypertension associated with HFpEF (PH-HFpEF), with a registrational path built around the LEVEL Phase 3 program and a planned second global Phase 3 study (LEVEL-2) accepted by FDA in 2025.
SANA Biotechnology
SANA Biotechnology is often framed as a high-risk / high-reward cellular engineering story. The core thesis revolves around its proprietary hypoimmune (HIP) platform: engineered cells designed to evade immune rejection, with the long-term goal of enabling allogeneic cell therapies without chronic immunosuppression.
VKTX Viking Therapeutics Inc
Viking Therapeutics is one of the most watched “metabolic” biotechs because it is trying to build a two-lane story: (1) obesity with VK2735 (the same molecule in injectable and oral tablet forms), and (2) MASH/NASH with VK2809 (thyroid receptor beta agonist with histology data).
BCRX BioCryst
(BCRX) Update: FDA Expands ORLADEYO Approval to Younger Children | Merlintrader trading Blog
ALT Altimmune Update dec 12

Altimmune announced a succession plan under which Jerry Durso will assume the role of President & CEO on January 1, 2026, while outgoing CEO Vipin Garg remains an advisor through June 30, 2026.
CYTK Cytokinetics
Cytokinetics is a late-stage cardiovascular biotech whose investment case now turns almost entirely around aficamten, a next-generation cardiac myosin inhibitor for obstructive hypertrophic cardiomyopathy (oHCM). The clinical package in oHCM is strong: the pivotal SEQUOIA-HCM trial versus placebo and the active-control MAPLE-HCM trial versus metoprolol both met their primary endpoints with clinically meaningful gains in peak oxygen uptake (pVO₂), improved symptoms and a safety profile that appears manageable so far.
ACIU AC Immune

AC Immune’s ACI-7104.056 is an active peptide vaccine designed to raise antibodies against misfolded α-synuclein in early Parkinson’s disease (PD). The ongoing Phase 2 VacSYn trial is the first randomized, double-blind study to show that such an approach can consistently drive very high antibody titers that reach the CNS and are associated with stabilization of multiple disease-relevant biomarkers.
DNLI Denali Therapeutics
Denali Therapeutics is a late-stage biotech focused on neurodegenerative and lysosomal storage diseases, with a proprietary Transport Vehicle (TV) platform to move biologics across the blood–brain barrier. The investment case in the next 12–18 months is dominated by the accelerated approval decision for DNL310 (tividenofusp alfa) in Hunter syndrome (MPS II), alongside the company’s ability to fund and execute a first commercial launch while advancing a broader pipeline.
FDA PDUFA Catalyst Report-January 2026
FDA PDUFA Catalyst Report January 2026 | Merlintrader trading Blog English Italiano FDA PDUFA Catalyst Report – January 2026 Report Catalysts FDA PDUFA – Gennaio 2026 Key biotech approval events with price scenarios and qualitative sentiment for active traders. Principali…
ATRA Atara Biotherapeutics Inc JAN 2 Update
Atara Biotherapeutics is a small-cap cell therapy company whose equity story is now essentially a single binary catalyst: the U.S. FDA decision on tab-cel (tabelecleucel) for EBV-positive PTLD, with a Priority Review PDUFA action date on 10 January 2026. The company has already divested or discontinued its other programs and has transferred most tab-cel operational responsibilities to Pierre Fabre Laboratories. What remains for shareholders is a high-volatility, high-uncertainty exposure to the outcome of that regulatory event and its knock-on impact on liquidity and solvency.
DAL Delta Air Lines
Delta today looks like the carrier you used to book for demanding but pragmatic business clients. Strong hub positioning in Atlanta, a wide global network and a product built around a rich mix of premium cabins and corporate traffic.
ALDX Aldeyra Therapeutics Update dec 12
Aldeyra Therapeutics is a clinical-stage biotech focused on inflammation, oxidative stress and ocular / metabolic diseases via its RASP platform. The lead asset, Reproxalap, is currently under FDA review for dry eye disease with a target action date of December 16, 2025 — a highly binary event coming after previous Complete Response Letters (CRLs) in 2023 and 2025.
OMER The Path of the Force Update dec 9

Omeros Corporation is a US-based biotechnology company focused on complement-related diseases. Its lead asset, narsoplimab (OMS721), targets transplant-associated thrombotic microangiopathy (TA-TMA), with a PDUFA target action date of December 26, 2025 for its resubmitted BLA. This is the core binary catalyst currently driving the stock.
SNTI Senti Biosciences Inc
Senti Biosciences is a micro-cap cell therapy company built around programmable gene-circuit technology. The lead asset, SENTI-202, is an off-the-shelf, logic-gated CAR-NK therapy in Phase 1 for relapsed / refractory acute myeloid leukemia (R/R AML).
VOR BIOPHARMA
VOR transformed from a dying cell therapy company into a focused autoimmune player with a globally licensed, China-approved dual BAFF/APRIL inhibitor (telitacicept). The stock has crashed/rallied 5 times in 2025 alone.
XCUR Exicure Inc
Exicure has repositioned itself around burixafor, a CXCR4 antagonist designed to mobilize hematopoietic progenitor cells (HPCs) for autologous stem-cell transplant in multiple myeloma and potentially other settings. At the ASH 2025 annual meeting, the company presented Phase 2 data showing high rates of successful HPC collection, including in daratumumab-exposed patients who are traditionally harder to mobilize.
PHAR Pharming Group (Updated dec 19)
Pharming Group has quietly moved into an unusual position in the biotech universe: it is a profitable, cash-generating rare-disease company, yet still trades in the small/mid-cap bracket and continues to carry several meaningful catalysts.
AQST Aquestive Therapeutics
Aquestive Therapeutics is a small-cap specialty pharma player built around its PharmFilm oral film technology and a mix of manufacturing revenue and proprietary pipeline assets. The stock is now tightly linked to a single binary catalyst: the FDA decision on Anaphylm, its epinephrine sublingual film for severe allergic reactions.
TVTX Travere Therapeutics
Travere Therapeutics is a commercial-stage rare-disease biotech focused on kidney and metabolic disorders. Its lead product, FILSPARI (sparsentan), is approved in the United States for IgA nephropathy (IgAN) and is under FDA review for focal segmental glomerulosclerosis (FSGS). A supplemental NDA for FSGS has an FDA target action date in early 2026, turning TVTX into a classic binary catalyst name with an already meaningful revenue base from IgAN.
FULC Fulcrum Therapeutics
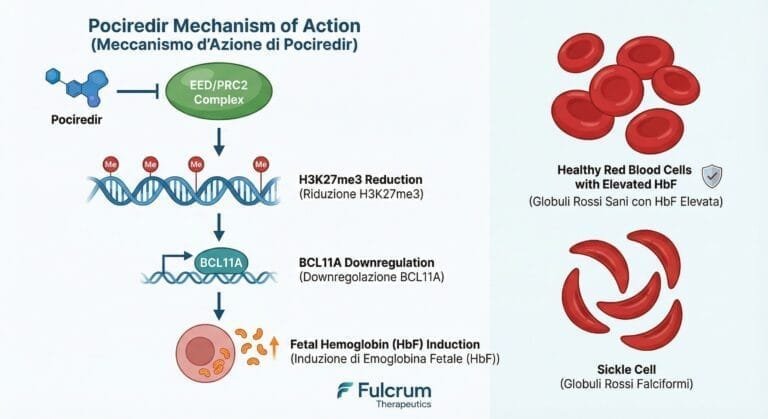
Fulcrum Therapeutics (FULC) – Pociredir SCD Deep Dive | Merlintrader trading Blog EN IT Fulcrum Therapeutics (FULC) – Pociredir in Sickle Cell Disease Deep Dive (December 2025) Last updated: December 8, 2025 – Authors: Merlintrader and Jane – Educational, non-promotional…